
- Institution: Stanford Univ Med Ctr Lane Med Lib/Periodical Dept/Rm L109
- Sign In as Member / Individual
Localization of Receptor-Mediated Signal Transduction Pathways: The Inside Story
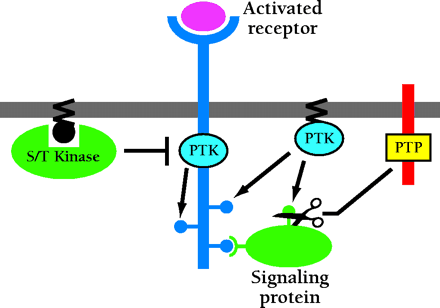
Compartmentalized regulation of receptor activities. Receptor-specific properties such as the pattern of receptor and signaling protein phosphorylation can be altered by endosome localization in a number of ways. Differential recruitment and/or activation of serine-threonine (S/T) kinases, which can phosphorylate the EGFR to inhibit its protein tyrosine kinase (PTK) activity, could change the extent and perhaps the specificity of substrate phosphorylation. Trafficking of other membrane-associated PTKs and receptor protein tyrosine phosphatases (PTPs) can yield higher or lower concentrations of these enzymes in endosomes versus the plasma membrane and provide additional regulation.


