Cancer Stem Cells: The Seeds of Metastasis?
Cancer begins as localized disease that will, if left untreated or ineffectively treated, metastasize to other organ sites. The origins of cancer and the process by which cancer can metastasize have been under investigation for decades, but have remained elusive. Recently, there has been a flurry of research regarding cancer stem cells (CSCs) and the role that these cells play in the initiation, progression and metastasis of cancer.
CSCs are a rare subpopulation of cancerous cells that are defined by their ability to give rise to tumors and the heterogeneous cells found within the tumor. CSCs were first observed in hematological malignancies (1) but have now been identified in solid tumors of breast (2), prostate (3, 4), brain (5), colon (6), and pancreas (7, 8). The most extensively studied CSCs include those from breast, prostate, and pancreatic tumors. CSCs are thought to be resistant to conventional chemotherapies and that this inherent resistance is what leads to relapse in many cancer patients (9) (Box 1). Indeed, Hermann et al. demonstrated that pancreatic CSCs are resistant to gemcitabine, the standard chemotherapeutic drug used in the treatment of pancreatic cancer (8). Therefore, CSCs are likely to survive conventional chemotherapies, result in patient relapse, and seed metastasis.
Cancer Stem Cells and Resistance to Conventional Chemotherapy
Certain properties of cancer stem cells (CSCs)—namely, their largely quiescent nature and their abundant expression of drug transporters (9)—support the hypothesis that CSCs are resistant to che-motherapeutic strategies designed to target rapidly dividing cells. To date, this hypothesis is gaining further evidence and documentation. For example, acute myeloid leukemia stem cells exhibit resistance to the conventional chemotherapeutic agent Ara-C (20) and pancreatic cancer stem cells are resistant to gemcitabine (8).
Metastasis of tumors has been linked with the ability of cells to undergo epithelial-mesenchymal transition (EMT). EMT and the ability to invade a basement membrane was first observed by Boyer et al. while studying rat bladder carcinoma cells (10). Since the publication of this study, the role of EMT has been established both in the morphogenesis of normal breast tissue (11) and the in vivo metastasis of many different cancer cells lines that were induced to undergo EMT in vitro [reviewed in (12)]. In order to metastasize, a neoplastic cell must first invade through the tissue (presumably through EMT), enter into the circulatory system, circulate, exit into a secondary tissue and establish a new tumor. If the only cells capable of giving rise to a tumor are CSCs, there must be a link between CSCs and metastasis. Indeed, CSCs are thought to be the cells that metastasize (13), and recently, a subset of pancreatic CSCs was observed to metastasize to bone when orthotopically transplanted in athymic mice (8). Also, breast CSCs were shown to be invasive through Matrigel (14)––a cell-culture scaffolding medium used to mimic more closely the in vivo extra-cellular compartment and used as an indicator of cellular meta-static potential––a process that relies on EMT. Thus, evidence is beginning to mount that CSCs are the cells capable of initiating metastasis.
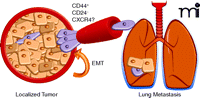
In their recent paper, Mani and colleagues demonstrate a surprising link between EMT and cancer stem cells, whereby non-stem mammary cells undergoing EMT dedifferentiate and obtain stem cell-like properties (15). Specifically, they show that by inducing EMT in immortalized human mammary epithelial cells (HMLEs), either by treating the cells with transforming growth factor–beta or by overexpressing the key transcription factors that control EMT (i.e., Snail and Twist), nearly all the cells acquire the CD44+CD24low phenotype, the phenotype previously identified for both normal breast stem cells (16) and breast CSCs (2). The stem-like CD44+CD24low cells isolated from HMLEs were able to differentiate into basal and luminal cells and formed secondary mammary structures when cultured in Matrigel, indicating that they did have properties of stem cells. Moreover, Mani et al. show that the increase in numbers of the CD44+CD24low cells did not merely represent an expansion of these cells but rather a conversion of the CD44lowCD24high cells to CD44+CD24low cells, an apparent dedifferentiation of the cells to a stem-cell phenotype. Mani et al. further show that enforced overexpression of either Snail or Twist in the HMLE cells–– transformed with a V12H-Ras oncogene to make them tumorigenic (HMLER cells)––resulted in tumor formation with as little as 100 cells, whereas parental HMLER cells not expressing Snail or Twist were nontumorigenic at this low cell number. This suggests that expression of either Snail or Twist, and subsequent EMT, results in an increase in the number of tumor-initiating CSCs. Their results show that the earliest step of metastasis (EMT) is capable of producing cells that have the ability to initiate a tumor and suggests that it may not be the existing CSCs that metastasize but rather cells that have reverted to this phenotype (Figure 1⇓). A direct study, however, demonstrating metastasis of these cells was not performed.
There have been only a few studies that examine the meta-static potential of CSCs, but collectively they suggest that the CSC phenotype alone is not enough to determine metastasis. In pancreatic cancer, CSCs (identified as CD133+ cells) were not able to metastasize when injected orthotopically at low numbers (8); however, a subset of CSCs, specifically those cells coexpressing the CSC marker CD133 and CXCR4 (i.e., CD133+CXCR4+), did metastasize to the liver when they were injected in low numbers. In mammary carcinomas, whereas CD44+CD24low cells were invasive through Matrigel (in vitro), the phenotype was not sufficient for metastasis (14) when cells were injected intracardiacally (in vivo). CXCR4 is also expressed on cells isolated both from bone and from lung metastases of breast carcinoma (17), but there are no studies that specifically determine if CD44+CD24− CXCR4+ breast cells are metastatic. Nonetheless, the acquisition of the CSC phenotype and tumor-initiating properties may, by itself, be insufficient to result in metastases. It is noteworthy, however, that the number of breast CSCs present within a tumor is predictive of distant metastasis (18). Although in the majority of mammary tumors studied (78%) there was a low prevalence of CSCs (≤ 10%), there was a very large proportion (80%) of CD44+CD24− cells present in a few cases. Could the large number of CSCs found within these tumors indicate that CSCs have been generated by EMT occurring within the breast tissue and that this occurrence is predictive of metastasis? Further studies combining markers of CSCs and EMT should provide insight into whether this is the case.
Because EMT is involved in the initial stages of metastasis, the generation of CSCs through this process might explain how distant sites are colonized. The relative low abundance of CSCs in breast tumors (18), coupled with the multiple and inefficient steps required to establish a metastatic tumor (19), would make the process of metastasis seem an unlikely event. Therefore, the ability of EMT to generate CSCs would make this process more efficient by ensuring that the cells that leave the site of the primary tumor are able to colonize other distant locations. Further studies of CSCs generated by EMT will need to be performed to determine if these CSCs can metastasize and evaluate if there is a subset of metastatic EMT-generated CSCs.
Proposed mechanism for the metastasis of breast cancer cells. Cells leaving a localized tumor undergo epithelial-mesenchymal transition (EMT) (indicated by the arrow) and acquire the characteristics of cancer stem cells thereby obtaining the ability to establish distant metastases, for example lung metastases. Epithelial cells are shown in pink, cancer stem cells are depicted in purple.
Acknowledgments
This research was supported in part by the Intramural Research Program of the NIH, National Cancer Institute, Center for Cancer Research. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
- © American Society for Pharmacology and Experimental Theraputics 2008
References

Elaine M. Hurt, PhD, obtained her degree from the University of Minnesota where she studied estrogen receptor–mediated transcriptional responses. Subsequent post-doctoral work in the laboratory of Louis M Staudt, MD, PhD, at the National Cancer Institute in Bethesda focused on profiling gene expression in lymphoma and multiple myeloma and understanding the role of c-maf in the etiology of myeloma. Currently, she is a staff scientist in the laboratory of Dr. Farrar where her studies are concentrated on understanding self-renewal and differentiation of prostate cancer stem cells. E-mail: hurte{at}ncifcrf.gov; fax 301-846-6104.

William L. Farrar, PhD, obtained his degree from the Virginia Polytechnic Institute. His postdoctoral work focused on cytokines and their signaling. Currently, he is the head of the Cancer Stem Cells Section in the Laboratory of Cancer Prevention at the National Cancer Institute in Frederick, MD where his studies are now focused on understanding the underlying biology of cancer stem cells. Using an integrated molecular profiling approach, he and his colleagues are investigating the mechanisms underpinning cancer stem cell differentiation, self-renewal, and invasion. E-mail: far-rar{at}mail.ncifcrf.gov; fax 301-846-6104.