NAADP: A New Second Messenger Comes of Age
One of the hallmarks of signal transduction by Ca2+ is complexity, which is mostly a consequence of the multiple, independent Ca2+-release mechanisms within cells. Following external stimulation, Ca2+ channels in the plasma membrane and on cellular organelles are activated in a specific spatio-temporal pattern to ensure the precise control of local, and eventually global, increases in concentrations of intracellular Ca2+ (1). The discovery of 1,4,5-trisphosphate (InsP3) as an intracellular Ca2+ mobilizer has led to a better understanding of the phospholipase C (PLC)–InsP3-mediated cellular signaling (2). Nicotinic acid adenine dinucleotide phosphate (NAADP) is the newest entry to the list of Ca2+-regulating second messengers (3). In NAADP, the nicotinamide ring in the molecule nicotinamide adenine dinucleotide phosphate (NADP) is replaced by nicotinic acid. NAADP mobilizes Ca2+ from stores that are now known to be important in a number of physiological processes, the common denominator of which is the interplay of the Ca2+ stores operated by the three Ca2+-linked second messengers NAADP, cyclic adenosine diphosphate ribose (cADPR), and InsP3. The stores activated by InsP3 and by cADPR are most likely the same: the sarco/endoplasmic reticulum. By general consensus, the stores operated by cADPR coincide with that mobilized through the ryanodine receptors (RyRs). Ever since its discovery as a Ca2+-mobilizing molecule in sea urchin egg homogenates, NAADP has exhibited a distinct pharmacological profile (4). Depletion of Ca2+ stores from endoplasmic reticulum (ER) in sea urchin homogenates or in intact eggs through the use of thapsigargin, a sarcoplasmic/ER calcium ATPase (SERCA) pump inhibitor, does not prevent NAADP-mediated release of Ca2+ from these preparations, suggesting that the NAADP-sensitive stores are not located in the ER. These findings are also in line with the distribution profile of NAADP-mediated Ca2+ stores, which do not co-migrate with those of the ER in cell fractionation experiments (5).
The most interesting finding in the early studies of NAADP has been its crosstalk with the other two well-characterized messengers of Ca2+ flux. Thus, Ca2+ release induced by NAADP elicits long-term Ca2+ oscillations mediated by the InsP3 receptor (InsP3R) and the cADPR/ryanodine receptor, indicating that a triggered Ca2+ release (by NAADP) is subsequently amplified by the two other messengers (6–9). Other unique properties of the NAADP-sensitive Ca2+ stores include the lack of inhibition by known modulators of InsP3R or RyR and a self-inactivation mechanism by subthreshold concentrations of NAADP that are, per se, insufficient to release Ca2+ (10). This auto-inactivation could reflect the presence of high and low affinity-binding sites in the NAADP receptors whereby a high affinity site mediates channel inactivation, and a low affinity site promotes channel opening. The auto-inactivation mechanism is also relevant to the concept of cross-talk among the three types of Ca2+ stores because the desensitization of NAADP-mediated Ca2+ release specifically blocks both InsP3 and cADPR-mediated signaling in T lymphocytes (11) and blocks cholecystokinin-induced Ca2+ oscillations in the apical pole of pancreatic acinar cells and the subsequent propagating Ca2+ wave (12). Additionally, the prior depletion of ER Ca2+ stores with thapsigargin and inhibition of RyRs with ryanodine inhibits the formation of NAADP-induced Ca2+ waves, further reinforcing the idea of mutual interaction of the three Ca2+ stores (13). But, if the NAADP-sensitive Ca2+ stores are not in the ER, where are they? Recent hints have come from copurification of a putative NAADP-sensitive store in a cell lysate fraction enriched with lysosomal markers and from the related observation that NAADP-induced Ca2+ release is inhibited by targeting drugs to lysosomes (14). These findings suggest that NAADP releases Ca2+ through a mechanism that utilizes a lysosomal-like organelle. However, this suggestion might not apply to all NAADP-sensitive stores—of which there could be more than one—or might not hold true for NAADP-sensitive stores in all cells. For instance, NAADP might directly modulate RyRs (15), possibly through an accessory protein, as shown by the opening of purified RyRs reconstituted in lipid bilayers (16). However, the finding that preparations of isolated pancreatic nuclei devoid of lysosomes or lysosomal-like acidic granules can still release Ca2+ into the nucleoplasm when challenged with NAADP is hard to reconcile with the idea that the release of Ca2+ by NAADP should originate from a Ca2+ store different from that of the ER (17).
The precise spatial distribution of NAADP receptors is essential for the mechanisms that underlie fertilization in starfish oocytes—an ideal system in which to study the details of Ca2+ homeostasis. The mechanism of egg activation has been studied intensively for more than a century and Ca2+ has been known for quite a while to play a vital role in it (18). When a sperm fertilizes an egg, it triggers a local explosive rise in intracellular Ca2+ concentrations that is then translated into a wave that sweeps through the egg. NAADP-activated and InsP3-activated receptors have recently been explored in detail in the starfish as determinants of the spatiotemporal dynamics of Ca2+ signaling during fertilization. In oocytes, the Ca2+ wave that follows the interaction with the sperm is preceded by a flash located in the cortical domain of the oocyte (cortical flash) generated during depolarization by the influx of Ca2+ through voltage-dependent plasma membrane channels (19). Injection of NAADP into the oocytes also induces a cortical Ca2+ flash, which then spreads throughout the cytoplasm as a wave (Figure 1A). A key point is that in starfish oocytes, the cortical flash is dependent on external Ca2+ (20, 21). The cortical flash begins with a localized Ca2+ increase, originating from a thapsigargin-sensitive NAADP-dependent store, the emptying of which activates an inwardly-directed Ca2+ current—movement of extracellular Ca2+ into the intracellular space—with properties similar to those of the current triggered by the sperm (Figure 1B) (22). Thus, the roles of NAADP-mediated and InsP3–mediated receptors in controlling the spatiotemporal dynamics of Ca2+ signaling during fertilization, previously assumed to be exclusively under the control of InsP3R, have now been separated and provide compelling evidence that NAADP triggers the initial circumscribed Ca2+ release that initiates the sperm-induced cortical flash, whereas InsP3Rs are involved in the later propagation of the Ca2+ wave in a Ca2+-induced Ca2+ release (CICR)–type mechanism (21). Supporting this conclusion are the findings that NAADP elicits the fertilization potential in starfish oocytes, and that the desensitization of NAADP Ca2+ stores prevents the onset of this fertilization potential (23).
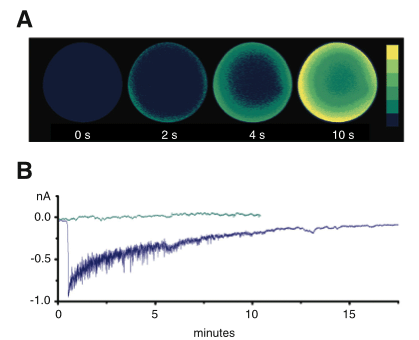
Ca2+ increase and membrane current activated by NAADP uncaging in a starfish oocyte. A. Pseudocolor relative fluorescent images of the Ca2+ increase induced by the global photoactivation of previously injected caged NAADP. NAADP induces a cortical flash that spreads centripetally to the oocyte center, which is dependent on extracellular Ca2+. B. Ca2+-mediated membrane current activated by the light-mediated liberation of NAADP. The current is affected by the preincubation with thapsigargin and injection of BAPTA [1,2-Bis(2-aminophenoxy)-ethane-N,N,N′,N′-tetraacetic acid] (22).
Different results had been instead initially obtained in intact sea urchin eggs, where the Ca2+ response to NAADP, it had been claimed, was independent of extracellular Ca2+ (7). More recent findings, however, indicate the response involves, as in starfish oocytes, a cortical flash produced by Ca2+ entry that is inhibited by the prior injection of NAADP to desensitize the NAADP receptors (24). That NAADP participates in fertilization is also supported by the demonstration that high (micromolar) levels of NAADP are also synthesized in sea urchin spermatozoa that have been activated by seawater (25).
Returning to the role played by NAADP in the fertilization process, it has been suggested that the NAADP-sensitive store activated by the sperm in sea urchin eggs is located in the lysosomes. This suggestion, however, is hard to reconcile with the finding that the NAADP-evoked current in starfish oocytes is not affected by lysosomal inhibitors such as bafilomycin, the vacuolar proton pump inhibitor, and by lycylphenylalanine 2-naphthylamide (GPN), which disrupts lysosomes through osmotic lysis. Bafilomycin, has also been tested in pancreatic acinar cells where it has no effect on the response to NAADP (26). Bafilomycin, however, does eliminate the NAADP-stimulated Ca2+ wave in pulmonary artery smooth muscle cells (27). Thus, much remains to be learned on the nature of the Ca2+ stores activated by NAADP.
Solving the controversies surrounding the mechanism of action and the spatiotemporal aspects of the function of NAADP is likely to take some time. Apart from the points alluded to above, additional issues need to be addressed. NAADP-induced Ca2+ release has now been described in many systems, including humans, but only recently it has been shown that the concentrations of NAADP in cells are controlled by physiological stimuli (8). Increased amounts of NAADP have been reported in pancreatic beta cells stimulated with glucose (28), and micromolar levels of NAADP have been found in activated sea urchin spermatozoa (25), but the signaling pathway leading to NAADP production is still obscure. CD38, a mammalian membrane bound ectoenzyme that catalyzes the synthesis of cADPR, might be involved in the pathway (8). It is noteworthy that CD38 is not exclusively expressed on the cell surface but it is also present in the cytoplasm (29–31) and can be internalised (32). Several tissues, including liver, heart, spleen, lung, and pancreas from CD38-knockout mice are unable to synthesize cADPR and NAADP (33). More information is needed, however, to understand the role played by CD38 and by intracellular soluble ADP-ribosyl cyclases in the regulation of intracellular Ca2+ concentrations (via cADPR and NAADP). Adding to this conundrum is the fact that the optimal pH of the reaction is out of the physiological range (34). The pH optimum issue, when coupled to the compartmentalization of nicotinic acid and NADP in an acidic environment such as that of the lysosomes, could strengthen the hypothesis of a lysosomal-like organelle as the NAADP-sensitive Ca2+ store (14). Nevertheless, other possible routes for NAADP synthesis in vivo should be explored, such as deamination of NADP or the phosphorylation of NAAD (35).
The molecular identity of the NAADP-sensitive receptors is still obscure, but a radioreceptor assay in sea urchin eggs, together with pharmacological studies, have revealed interesting features in the NAADP binding site. Very importantly, NAADP binding seems to be irreversible, suggesting an explanation for the molecular basis of the inactivation phenomenon (36–38). Whatever their nature, NAADP receptors display peculiar features that make them distinct among effectors of Ca2+ stores. Their identification will clearly be an important achievement. Finally, after all that has been discussed here is clarified, one exceptionally important point will have to be addressed: the molecular mechanism by which NAADP cross talks to the receptor molecule(s), wherever they are.
- © American Society for Pharmacology and Experimental Theraputics 2005
References

Luigia Santella, PhD, is a senior scientist and group leader at the Cell Signaling Laboratory, Stazione Zoologica “Anton Dohrn”, Naples, Italy. Her research focuses on calcium signaling during the meiotic cycle (maturation) and fertilization of starfish oocytes. E-mail santella{at}szn.it; fax +39-081-7641355.



