 |
 |

Chromium: Friend or Foe?
David J. Porter, MD;
Lawrence W. Raymond, MD, ScM;
Geraldine D. Anastasio, PharmD, BCPS
Arch Fam Med. 1999;8:386-390.
ABSTRACT
Chromium is an essential nutrient with many natural sources, but several investigators have presented evidence that healthy persons often have an inadequate intake of this metal. Advocates of its use as a dietary supplement believe that people with diabetes, lipoprotein abnormalities, and increased risk of cardiovascular disease may benefit from such supplementation. Others suggest a therapeutic role for chromium in obese people and in those who seek to improve their body composition for other reasons, but research provides little support for these uses. For the general public, current data do not warrant routine use of chromium supplements, whose risk-benefit function has not yet been adequately characterized.
INTRODUCTION
An upsurge of public interest in dietary supplements has contributed to debate in the medical community regarding the value of "alternative therapies," for which the National Institutes of Health has established a special office.1 Such therapies include dietary supplements, which, according to one study,2 were being used by 53% of family practice patients queried. Family physicians are striving to become more informed about these and other forms of complementary and alternative medicine3 to counsel patients appropriately about their use. A supplement gaining the attention of the popular press and medical literature4 is chromium. In this article we summarize what is known about this element as a nutritional supplement based on recent English-language articles and concentrate on its possible therapeutic value and potential adverse effects. Of more than 3000 citations about chromium published since 1990, we reviewed more than 100 articles describing possible beneficial effects and adverse outcomes of chromium supplementation. We did not attempt to cover all of the recent literature but aimed instead to provide family physicians with a balanced update on this emerging area of investigation.
CHROMIUM VALENCE DETERMINES BIOLOGIC ACTIVITY
Chromium is a fascinating and versatile metal discovered 2 centuries ago. It makes the emerald green and the ruby red. An essential trace nutrient, its biologic activity depends on its valence state. Metallic chromium (valence zero, Cr-0) is inert. Of the 2 other stable forms, chromium-III (Cr-III) may be of therapeutic value and thus is the main focus of this article. It is the form present in foodstuffs and in chromium picolinate, which is used as a nutritional supplement because of its better bioavailability.
Chromium-VI (Cr-VI) is the form found in the welding, plating, and chemical industries, where it is a recognized respiratory irritant and carcinogen.5 The carcinogenic action of Cr-VI requires its intracellular reduction to Cr-III.5-7 Although intranuclear Cr-III may thus be considered the proximate carcinogen for Cr-VI, no carcinogenic activity has been ascribed to ingested Cr-III.7-8 However, Cr-III has not been adequately evaluated in this regard, perhaps because of its limited ability to cross cell membranes.5, 7
Intranuclear penetration has generally been thought to be limited to Cr-VI. On the other hand, results of some recent studies9-11 raised concerns about the safety of Cr-III as a long-term nutritional supplement because of possible genotoxic effects. One authority12 expressed the view that all forms of chromium (including Cr-III) should be considered human carcinogens, although this view has been challenged.13-14 The International Agency for Cancer Research considered the question moot in 1980,15 stating that Cr-III was "unclassifiable" on the basis of then-existing evidence (see the "Questionable Uses, Adverse Effects, and Contraindications" section).
SOURCES AND ADEQUACY OF CHROMIUM INTAKE
Bioavailable Cr-III salts are found in many foodstuffs, especially liver, American cheese, brewer's yeast, and wheat germ. Many meats, fish, fruits, whole grains, and vegetables (eg, carrots, potatoes, and spinach) are also good sources, as are alfalfa, brown sugar, molasses, and animal fats.5 Normal diets in the United States provide about 15 µg of Cr-III per 4.2 kJ (1000 calories) of intake,16 but little of this is absorbed depending on what form is ingested. No recommended dietary allowance or Food and Drug Administration–approved indications for chromium supplementation have been published, but the National Research Council estimates that intakes of 50 to 200 µg/d are safe and adequate.17 The National Research Council estimates that Cr-III intake in the United States is 25 to 33 µg/d, with more than 90% of persons having a daily intake of less than 50 µg/d.
The frequency of actual chromium deficiency in the general population is unknown, absent a reliable indicator of the adequacy of Cr-III nutriture. Several cases of subacute syndromes of Cr-III deficiency have been reported in patients receiving total parenteral nutrition.18 Inadequate dietary intake also has been implicated in several metabolic abnormalities, including impaired glucose and lipid metabolism, elevated circulating insulin levels, and decreases in insulin-receptor numbers.
Chromium-III repletion after experimental dietary depletion improved glucose tolerance and reversed abnormal elevations in circulating insulin and glucagon in patients with mild hyperglycemia.18 Intake of Cr-III also has been shown to improve glycemic control and cholesterol levels in Chinese patients with type 2 diabetes, whose pretreatment status with regard to this supplement or other nutrients was not described.19
AVAILABILITY AND POSSIBLE USES OF CHROMIUM SUPPLEMENTS
Chromium-III is available in the chloride or picolinate salt form; it also has several naturally occurring sources in organic complexes with nicotinic acid.8 Most recent studies of chromium therapy used Cr-III doses of 200 to 1000 µg/d in the picolinate or chloride form, similar to manufacturers' advice. A month's supply of 200-µg tablets of Cr-III picolinate may cost as little as $2 to $5, whereas 100 capsules of 500 µg each cost about $9 from a health food catalog. Brewer's yeast is said to provide 30 µg of readily absorbed Cr-III per 1.6 g20-21 and costs about $8 for 100 tablets. These formulations have not been reported to cause toxic effects at the preceding doses.
In combination with nicotinic acid and amino acids, Cr-III forms a complex called glucose tolerance factor.20, 22 Although the structure of glucose tolerance factor has not been elucidated, it is known to enhance the peripheral actions of insulin, leading to the concept that people with diabetes might benefit from Cr-III supplementation.18-19 Those with heart disease or lipoprotein abnormalities might also be candidates for Cr-III supplements, in light of reports of beneficial effects on lipid metabolism,4, 8, 19 provided long-term safety concerns can be resolved. Although suggestions have been made that Cr-III supplements might also help preserve bone density and otherwise improve body composition, studies have yielded unconvincing results.
PHARMACOLOGY
Ingested chromium is poorly absorbed, eg, about 1% of Cr-III and 10% of Cr-VI.5, 23 When complexed to other ligands such as nicotinic acid and amino acids in glucose tolerance factor, Cr-III is 25-fold more readily absorbed; 8 to 10 µg/d may be an adequate intake in the form of brewer's yeast, for example. Most of what is known about the kinetics of Cr-III comes from whole-body and plasma counting studies by Lim et al24 using intravenous 51Cr. It circulates largely bound to transferrin in the blood. There is a little storage in the liver, spleen, fat, and bone, and more than two thirds of an absorbed ingested dose is rapidly excreted in the urine and bile. Plasma clearance occurs within 8 to 12 hours, and tissue elimination has a half-life of several days. The normal concentration of Cr-III in whole blood is 380 to 580 nmol/L. Most is contained in erythrocytes, plasma levels being only 1.9 to 5.8 nmol/L.25 This makes it technically difficult to use these levels in monitoring. Serum or urine levels do not reflect body stores of chromium,22 but urinary excretion of 10 µg/d is usual in the absence of oral supplementation or industrial exposure.23 The mechanisms underlying the effects of Cr-III on glucose and lipid metabolism are still being elucidated. These efforts rely mainly on effects of Cr-III deprivation and supplementation because of analytical difficulties and the inability otherwise to gauge the adequacy of chromium nutriture.
For those exposed to chromium industrially (eg, during welding or plating), Cr-VI is the valence form involved; it generally enters the body from such exposures by inhalation or transdermally and in this form readily crosses cell membranes. Once within the cellular milieu, Cr-VI is rapidly reduced to Cr-III, which has carcinogenic activity only within cell nuclei.
EVIDENCE OF EFFICACY
As reviewed by Mertz,22 most but not all of 15 controlled studies reported beneficial effects of Cr-III supplementation on insulin efficiency or blood lipoproteins. However, it is unclear which individuals will benefit from such Cr-III supplements. This remains a deterrent to their use, as do the incomplete understanding of the therapeutic mechanisms involved and the possible adverse effects of long-term use.
Diabetes
The strongest evidence that Cr-III supplementation might be beneficial in humans comes from studies by Anderson et al18 in patients with diabetes and in healthy subjects whose glucose tolerance was impaired experimentally by chromium depletion. In a more recent study,19 180 Chinese men and women with type 2 diabetes were given placebo or 200 or 1000 µg of Cr-III picolinate for 4 months. China was selected for the study to minimize the use of nutritional supplements. It is unclear whether the customary Chinese intake of Cr-III differs from that of Western diets, perhaps limiting wider applicability of these findings. These findings included Cr-III dose-related improvements in blood levels of glycated hemoglobin, fasting and 2-hour postprandial glucose, insulin, and total cholesterol. No changes were found in blood levels of triglycerides, high-density lipoprotein (HDL), or urea nitrogen or in body mass index. Four earlier controlled studies discussed by Finney and Gonzalez-Campoy16 found no beneficial effects of Cr-III supplementation in other patients with diabetes. A recent case report21 illustrates how individual patients with diabetes may decide for themselves to try Cr-III supplementation to help manage glucose control, sometimes encountering hypoglycemia in the process.26 The American Diabetes Association27 position as of 1996 was that "chromium supplementation has no known benefit" in patients with diabetes who are not chromium deficient. Use of Cr-III by patients with gestational diabetes was reported by Jovanovic-Peterson et al28 to have beneficial effects on glycated hemoglobin, insulin, and glucose levels.
Lipoproteins
The 1993 review by Mertz22 cited 4 positive and 4 negative studies of the effects of Cr-III intake on lipoprotein concentrations. Others have since examined the effects of Cr-III supplementation on serum lipoprotein levels in various patient groups. For example, Roeback et al29 reported a mean increase of 6 mg/dL in HDL concentrations of 35 male veterans taking 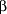 -adrenergic blocking agents for hypertension or coronary disease who took 600 µg/d of Cr-III as commercially supplied glucose tolerance factor for 8 weeks. There were no changes in total cholesterol level, triglyceride level, body weight, and HDL or other lipoprotein concentrations among 37 men who took placebo capsules. However, Lee and Reasner25 also found no change in total, low-density lipoprotein, or HDL cholesterol levels, or in glucose regulation, in Hispanic patients with diabetes given Cr-III picolinate, 200 µg/d, for 2 months. Triglyceride levels decreased 17% in the Cr-III–supplemented phase and not in the placebo crossover phase. Hendler8 described an improvement in lipoprotein balance in a controlled study using Cr-III picolinate, 200 µg/d, but this work has not been published. Simon4 stated that pending further study, it seems reasonable to give Cr-III supplements to patients who have low levels of HDL, but other pharmacological approaches currently have a more solid research base. -adrenergic blocking agents for hypertension or coronary disease who took 600 µg/d of Cr-III as commercially supplied glucose tolerance factor for 8 weeks. There were no changes in total cholesterol level, triglyceride level, body weight, and HDL or other lipoprotein concentrations among 37 men who took placebo capsules. However, Lee and Reasner25 also found no change in total, low-density lipoprotein, or HDL cholesterol levels, or in glucose regulation, in Hispanic patients with diabetes given Cr-III picolinate, 200 µg/d, for 2 months. Triglyceride levels decreased 17% in the Cr-III–supplemented phase and not in the placebo crossover phase. Hendler8 described an improvement in lipoprotein balance in a controlled study using Cr-III picolinate, 200 µg/d, but this work has not been published. Simon4 stated that pending further study, it seems reasonable to give Cr-III supplements to patients who have low levels of HDL, but other pharmacological approaches currently have a more solid research base.
QUESTIONABLE USES, ADVERSE EFFECTS, AND CONTRAINDICATIONS
Claims of efficacy for chromium supplementation in commercial health food and fitness publications include body building and postmenopausal maintenance of bone mass. However, little peer-reviewed research supports such claims, and some adverse effects have been reported.
Obesity and Body Composition
Beneficial effects of Cr-III supplementation on body composition and weight loss have been asserted by manufacturers.30 Although Evans31 reported improved fat-free body composition after 6-week supplementation with Cr-III picolinate, 200 µg/d, using skinfold measurements, results of 7 other studies32-38 showed no such beneficial effects. Grant et al38 reported that obese women taking Cr-III for weight loss actually gained weight unless they also exercised. Another group noted possible interference with iron transport, as found earlier in animal studies.37 Thus, the Federal Trade Commission in 1996 ordered 3 manufacturers to cease making unsupported claims of health benefits from Cr-III supplementation.30
Genotoxic Effects
As stated earlier, ingested Cr-III has not been shown to have carcinogenic activity.5, 7-8 It has not been adequately studied in this regard, however, perhaps because of its limited ability to cross cell membranes. Some recent studies raised concerns about possible genotoxic effects from prolonged use of Cr-III supplements. For example, evidence9, 39 now exists that Cr-III can enter the nuclei of liver and kidney cells of intact rats and mice and bind to DNA. It does so at a slower rate than Cr-VI, and actual DNA damage was produced only by Cr-VI and not by Cr-III. That is, with the methods used, Cr-III treatment did not cause the DNA cross-links or strand breakage produced by Cr-VI. Cupo and Wetterhahn9 initially expressed the view that DNA damage is generally less likely after Cr-III than Cr-VI treatment, based on differences in mutagenicity of these 2 valence forms. More recently, investigators from the same laboratory provided a basis for greater concern about the safety of Cr-III as a human nutritional supplement. First, Wetterhahn's group showed that use of Cr-III picolinate causes chromosomal damage in Chinese hamster ovary cells.10 In a companion article,11 using pharmacokinetic modeling, the authors indicated that prolonged intake of this form of dietary supplement may lead to tissue levels at which adverse effects occur in animals and in vitro.
Although rebutted in correspondence by the manufacturer,40 these results suggest the need for caution as to the safety of prolonged use of chromium picolinate.41-42 However, the preceding articles do not distinguish effects of Cr-III supplementation from those that might be caused by the picolinate moiety. The argument that genotoxic effects cannot occur because of Cr-III's limited ability to cross cell membranes is not compelling because some membrane permeation must be occurring for ingested Cr-III to be absorbed and bioactive in the first place.
As stated previously, most documented adverse effects of chromium have been related to Cr-VI encountered in industry.5, 23 However, as with other unregulated dietary supplements, there is concern that as use of Cr-III increases as a result of promotional efforts, more evidence of toxic effects may emerge. For example, a recent case report43 of acute renal failure caused by severe interstitial nephritis after 5 months of chromium picolinate intake (600 µg/d) sounds a cautionary note about such over-the-counter Cr-III supplementation. Another case report44 describes severe systemic illness in a 33-year-old woman who took 1200 to 2400 mg/d of chromium picolinate for 5 months. She developed hemolysis, thrombocytopenia, hepatic dysfunction, and renal failure but responded to hemodialysis. She had normal renal function and other laboratory values 1 year later. Other evidence of possible Cr-III nephrotoxicity comes from animal studies in which rats and rabbits were injected daily with 2 mg/kg for 3 to 6 weeks45 and also demonstrated hepatic injury.
Chromium-III caused fewer toxic effects by oral administration, probably because of its lesser bioavailability in the forms used.45 Consumer Reports (November 1995, pp 698-705) quoted the Food and Drug Administration as having concerns about cardiac irregularities, but most reports of adverse events to the Food and Drug Administration involved Cr-III taken in conjunction with various herbal preparations, ephedrine, or other pharmacological agents (Table 1), which may have been responsible for the adverse events.46 Of 6632 adverse events reported to the Special Nutritionals Adverse Events Monitoring System as of May 14, 1998, 527 involved chromium-containing products, but 503 of these reports concerned 74 multicomponent products. One product accounted for 150 of these adverse effect reports and contained herbs such as Ma-huang, cola nut, ginger, willow bark, ginkgo biloba, bladder wrack, fo-ti, hawthorn berry, saw palmetto, beet, and boron picolinate in addition to chromium picolinate and chelate. Only 23 products consisted of chromium picolinate alone.
|
|

|
|
Adverse Events Reported to the Food and Drug Administration Related to Supplementation With Chromium-Containing Special Nutritionals*
|
|
|
No contraindications to the use of chromium have been identified, but it seems prudent to monitor renal function if it is used in patients with known renal disease. Treatment of gestational diabetes16 with Cr-III has not been evaluated for fetal toxic effects or safety in nursing mothers or children. The relevance of fetotoxic effects from Cr-VI exposure reported by Junaid et al47 is unclear in this regard.
Most known adverse effects of industrial exposures to chromium—also related to Cr-VI rather than to Cr-III—have no direct relevance to the supplements we discussed. However, these effects include corrosive skin lesions, lung cancer, and kidney and liver toxic effects,5, 23 so they might be a source of confusion and concern to patients. The acute oral lethal dose of chromium is about 1 g, and chelation with British antilewisite may be most useful in treating severe overdoses,48 which thus far have not been published.
CONCLUSIONS
Chromium, like some other dietary supplements, has only a mixed research base to support the claims of its proponents. Its role in insulin regulation and its possible therapeutic effects in people with diabetes are among the more attractive prospects. Chromium supplementation has a beneficial effect on the relationship between glucose and insulin in Cr-III–deficient patients with diabetes, but most are not so deficient. We lack direct ways to assess chromium status in persons with insulin resistance and glucose intolerance. Thus, we cannot readily identify those most likely to benefit from chromium supplementation. Such supplementation might also improve lipoprotein balance in persons at risk for coronary disease. It is unclear whether improvement in the studies mentioned was a direct effect of chromium intake on lipid metabolism or an indirect consequence related to improved glucose and insulin homeostasis.
The manufacture and marketing of chromium is not controlled by the Food and Drug Administration, so there is no standardization in manufacturing or postmarketing surveillance. We also lack the ability to monitor treatment via blood levels or other means. Renal insufficiency seems to be a rare complication of therapy at doses higher than the 600 mg/d currently recommended. Nevertheless, it behooves family physicians and other health care professionals to be aware of the potential adverse effects and the possible benefits of this therapy. We must continue to watch for new research findings on Cr-III and to advise our patients accordingly.
Since the manuscript was accepted for publication, a note was added in proof: a case of acute rhabdomyolysis occurred in a 24-year-old female body builder who ingested 1200 µg of Cr-III picolinate over 48 hours.49
AUTHOR INFORMATION
Accepted for publication October 15, 1998.
Corresponding author: Lawrence W. Raymond, MD, ScM, Department of Family Medicine, Carolinas Medicial Center, PO Box 32861, Charlotte, NC 28232.
From the Departments of Family Medicine, Carolinas Medical Center, Charlotte, NC, and University of North Carolina at Chapel Hill.
REFERENCES
 |  |
1. Angell M, Kassirer JP. Alternative medicine: the risks of untested and unregulated remedies. N Engl J Med. 1998;339:839-841.
FREE FULL TEXT
2. Fliason B, Myszkowski J, Marbella A, Basmann D. Use of dietary supplements by patients in a family practice clinic. J Am Board Fam Pract. 1996;9:249-253.
3. Spencer J, Jonas W. And now, alternative medicine. Arch Fam Med. 1997;6:155-156.
FREE FULL TEXT
4. Simon H. Patient-directed, nonprescription approaches to cardiovascular disease. Arch Intern Med. 1994;154:2283-2296.
FREE FULL TEXT
5. Sawyer HJ. Chromium and its compounds. In: Zenz C, Dickerson OB, Horvath EP, eds. Occupational Medicine. St Louis, Mo: Mosby–Year Book Inc; 1994.
6. Snow ET. Effects of chromium on DNA replication in vitro. Environ Health Perspect. 1994;102(suppl 3):41-44.
7. Hathaway JA. Role of epidemiologic studies in evaluating the carcinogenicity of chromium compounds. Sci Total Environ. 1989;86:169-179.
FULL TEXT
| PUBMED
8. Hendler SS. The Doctors' Vitamin and Mineral Encyclopedia. New York, NY: Simon & Schuster; 1990:125-127.
9. Cupo DY, Wetterhahn KE. Binding of chromium to chromatin and DNA from liver and kidney of rats treated with sodium dichromate and chromium (III) chloride in vivo. Cancer Res. 1985;45:1146-1151.
FREE FULL TEXT
10. Stearns DM, Wise JP, Patierno SR, Wetterhahn KE. Chromium (III) picolinate produces chromosome damage in Chinese hamster ovary cells. FASEB J. 1995;9:1643-1649.
ABSTRACT
11. Sterns DM, Belbruno JJ, Wetterhahn KE. A prediction of chromium (III) accumulation in humans from chromium dietary supplements. FASEB J. 1995;9:1650-1657.
ABSTRACT
12. Norseth T. The carcinogenicity of chromium and its salts. Br J Ind Med. 1986;43:649-651.
ISI
| PUBMED
13. Petrilli FL, DeFlora S. Carcinogenicity of chromium and its salts [letter]. Br J Ind Med. 1987;44:355.
PUBMED
14. Levy LS, Martin PA, Venitt S. Carcinogenicity of chromium and its salts [letter]. Br J Ind Med. 1987;44:355-357.
15. International Agency for Research on Cancer. Chromium and chromium compounds. IARC Monogr Eval Carcinog Risk Chem Hum. 1980;23:205-323.
PUBMED
16. Finney LS, Gonzalez-Campoy JM. Dietary chromium and diabetes: is there a relationship? Clin Diabetes. Jan/Feb 1997:6-8.
17. National Research Council. Recommended Dietary Allowances. 10th ed. Washington, DC: National Academy Press; 1989.
18. Anderson R, Polansky M, Bryden N, Canary J. Supplemental chromium effects on glucose, insulin, glucagon, and urinary chromium losses in subjects consuming controlled low-chromium diets. Am J Clin Nutr. 1991;54:909-916.
FREE FULL TEXT
19. Anderson RA, Cheng N, Bryden NA, et al. Elevated intakes of supplemental chromium improve glucose and insulin variables in individuals with type 2 diabetes. Diabetes. 1997;46:1786-1791.
ABSTRACT
20. Mooradian A, Failla M, Hoogwerf B, Maryniuk M, Wylie-Rosett J. Selected vitamins and minerals in diabetes. Diabetes Care. 1994;17:464-479.
ISI
| PUBMED
21. Fox GN, Sabovic Z. Chromium picolinate supplementation for diabetes mellitus. J Fam Pract. 1998;46:83-86.
ISI
| PUBMED
22. Mertz W. Chromium in human nutrition: a review. J Nutr. 1993;123:626-633.
23. Kapil V, Keogh J. Chromium Toxicity: Case Studies in Environmental Medicine. Rockville, Md: Agency for Toxic Substances and Disease Registry, US Dept of Health and Human Services; 1990.
24. Lim TH, Sargent T, Kusubov N. Kinetics of trace element chromium (III) in the human body. Am J Physiol. 1983;244:R445-R454.
25. Lee N, Reasner C. Beneficial effect of chromium supplementation on serum triglyceride levels in NIDDM. Diabetes Care. 1994;17:1449-1452.
ABSTRACT
26. Porterfield LM. Sudden hypoglycemia in a diabetic. RN. 1996;59:71.
27. American Diabetes Association. Position statement: nutrition recommendations and principles for people with diabetes mellitus. Diabetes Care. 1996;19(suppl):S16-S19.
28. Jovanovic-Peterson L, Gutierrez M, Peterson C. Chromium supplementation for gestational diabetic women improves glucose tolerance and decreases hyperinsulinemia [abstract]. Diabetes Care. 1996;45(suppl 2):337A.
29. Roeback JR, Hla KM, Chambless LE, Fletcher RH. Effects of chromium supplementation on serum high-density lipoprotein levels in men taking beta-blockers. Ann Intern Med. 1991;115:917-924.
30. McArdle WD, Moore BJ. Chromium shows little proof as weight loss supplement. Altern Med Alert. 1998;1:9-10.
31. Evans GW. The effect of chromium picolinate on insulin controlled parameters in humans. Int J Biosoc Med Res. 1989;11:163-180.
32. Hasten DL, Rome EP, Franks BD, Hegsted M. Effects of chromium picolinate on beginning weight training students. Int J Sports Nutr. 1992;2:343-350.
ISI
| PUBMED
33. Clancy SP, Clarkson PM, DeCheke ME, et al. Effects of chromium picolinate supplementation on body composition, strength, and urinary chromium loss in football players. Int J Sports Nutr. 1994;4:142-153.
ISI
| PUBMED
34. Almada A, Kreider R, Harmon K, et al. Effects of ingesting a nutritional supplement containing chromium picolinate and boron on body composition during resistance-training [abstract]. FASEB J. 1995;9:A1015.
35. Trent LK, Thieding-Cancel D. Effects of chromium picolinate on body composition. J Sports Med Phys Fitness. 1995;35:273-280.
ISI
| PUBMED
36. Hallmark MA, Reynolds TH, Desouza CA, Dotson CD, Anderson RA, Rogers MA. Effects of chromium and resistive training on muscle strength and body composition. Med Sci Sports Exerc. 1996;28:139-144.
ISI
| PUBMED
37. Lukaski HC, Bolonchuk WW, Siders WA, Milne DB. Chromium supplementation and resistance training: effects on body composition, strength, and trace element status of men. Am J Clin Nutr. 1996;63:954-965.
FREE FULL TEXT
38. Grant KE, Chandler RM, Castle AL, Ivy JL. Chromium and exercise training: effect on obese women. Med Sci Sports Exerc. 1997;8:992-998.
39. Tandon SK, Saxena DK, Gaur JS, Chandra SV. Comparative toxicity of trivalent and hexavalent chromium. Environ Res. 1978;15:90-99.
PUBMED
40. McCarty MF. Chromium picolinate. FASEB J. 1996;10:365-366.
ISI
| PUBMED
41. Stearns DM, Wetterhahn KE. Chromium (III) picolinate. FASEB J. 1996;10:367-369.
42. Kortenkamp A. Pharmacokinetic modelling in chromium risk assessment. Hum Exp Toxicol. 1996;15:601-602.
ISI
| PUBMED
43. Wasser WG, Feldman NS, D'Agati VD. Chronic renal failure after ingestion of over-the-counter chromium picolinate [letter]. Ann Intern Med. 1997;126:410.
FREE FULL TEXT
44. Cerulli J, Grabe DW, Gauthier I, Malone M, McGoldrick MD. Chromium picolinate toxicity. Ann Pharmacother. 1998;32:428-431.
ABSTRACT
45. Katz SA, Salem H. The toxicology of chromium with respect to its chemical speciation: a review. J Appl Toxicol. 1993;13:217-224.
FULL TEXT
|
ISI
| PUBMED
46. The Special Nutritionals Adverse Event Monitoring System (SN/AEMS) web report. Available at: http://www.verity.fda.gov/search97. Accessed May 14, 1998.
47. Junaid M, Murthy RC, Saxa DK. Chromium toxicity in mice during late pregnancy. Vet Hum Toxicol. 1995;37:320-323.
ISI
| PUBMED
48. Merck Manual. 16th ed. Whitehouse Station, NJ: Merck & Co; 1992:2685.
49. Martin WR, Fuller RE. Suspected chromium picolinate-induced rhabdomyolysis. Pharmacotherapy. 1998;18:860-862.
ISI
| PUBMED
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 1999;8(5):383-385.
FULL TEXT
|