 |
 |

The Effect of Glycemic Control on the Incidence of Macrovascular Complications of Type 2 Diabetes
Michael P. Stern, MD
Arch Fam Med. 1998;7:155-162.
ABSTRACT
Of all the complications of diabetes mellitus, macrovascular complications, ie, large-vessel atherosclerosis, account for the largest share of morbidity, mortality, and health care expenditures. Whereas there is now highly persuasive evidence that glycemic control reduces the risk of microvascular complications in type 1 diabetes, and probably in type 2 diabetes as well, such evidence is unavailable for macrovascular complications. Prospective epidemiologic studies, however, indicate that poor glycemic control enhances cardiovascular risk, and a number of biochemical mechanisms have been advanced to explain this phenomenon. However, data from animal studies, in vitro studies, and prospective epidemiologic studies suggest that endogenous insulin or insulin resistance may be atherogenic. Thus, a dilemma exists for insulin treatment, although the weight of evidence still favors its aggressive use. For persons whose glycemia can be adequately controlled with oral agents, the use of agents such as metformin and troglitazone—which do not raise, and may even lower, insulin concentrations—may offer an advantage. Definitive clinical trials on the benefits and risks of insulin therapy related to macrovascular complications are lacking and urgently needed.
INTRODUCTION
From a public health perspective, the macrovascular complications of diabetes head the list of complications in causing morbidity, mortality, and health care expenditures. Roughly 40% of total mortality in persons with diabetes is caused by ischemic heart disease, with other categories of cardiovascular disease (CVD), including stroke, accounting for an additional 25%.1 The next leading cause of death in persons with diabetes, malignant neoplasms, causes only 13% of total deaths.1 For morbidity, data from the National Hospital Discharge Survey indicate that, of the 154.5 per 1000 patients with diabetes hospitalized in 1992 for the treatment of complications, 58% were for the treatment of CVD.2 Only about 10% were for the treatment of renal complications. Although nontraumatic amputations are almost 12 times more common in persons with diabetes than in persons without diabetes, in absolute numbers they accounted for fewer than 5% of 1992 hospitalizations for diabetic complications.2 These figures highlight the overwhelming primacy of CVD in producing diabetic morbidity.
In general, the risk of cardiovascular mortality is from 2 to 4 times higher in persons with diabetes than in persons without diabetes.1 Several studies have indicated that diabetes increases cardiovascular risk more in women than in men, thereby eliminating, either wholly or in part, the female protection against coronary disease that is observed in the general population. In the Framingham Study, for example, the age-adjusted risk of dying of CVD was 2.1 times higher in men with diabetes than in men without diabetes, but for women, this relative risk was 4.9.3 The corresponding relative risks in the Rancho Bernardo Study in southern California were 1.8 for men and 3.3 for women.4 Not all studies, however, show this pattern. In a recent summary of 16 studies of mortality in persons with diabetes,1 a higher relative risk for CVD in women compared with men was observed in 8 studies, and 2 others showed this pattern in younger, but not in older, women. In the remaining 6 studies, the relative risks for CVD in persons with diabetes vs persons without diabetes were relatively similar in the 2 sexes.
Because of the aging of the US population, these findings have great public health relevance. Combined with the aging of the population and the greater longevity of women compared with men, a loss of protection against coronary disease among women with diabetes would markedly increase the absolute number of future cases of CVD among women. Minorities are also a growing portion of the US population, and increased rates of diabetes have been documented among African Americans,5 Hispanics,6 and Native Americans.7 It has long been believed that, despite their high rates of diabetes, CVD was rare in Native Americans. This conclusion has been heavily influenced by data on Pima Indians, among whom CVD is rare.8 The Strong Heart Study, however, has shown that, although CVD is relatively less common in Native Americans from the southwestern United States, this is not necessarily the case for all Native Americans. Native Americans from Oklahoma and North and South Dakota, for example, showed high rates of CVD.9 It also has been claimed that Mexican Americans have lower CVD mortality than non-Hispanic whites,10 but this conclusion, too, has been challenged.11 For example, in the Corpus Christi Heart Project, the rate of admission to coronary care units for the treatment of myocardial infarction was higher among Mexican Americans than among non-Hispanic whites.12 These data suggest that in future years, the number of cases of CVD among persons with diabetes from minority populations is likely to increase.
Many studies have indicated that the increased cardiovascular risk associated with diabetes is not merely the result of increased levels of conventional cardiovascular risk factors, eg, dyslipidemia, hypertension, and obesity.1 Figure 1 shows the 12-year cardiovascular mortality among almost 350000 men screened for entry in the Multiple Risk Factor Intervention Trial, approximately 5000 of whom had diabetes when first screened.13 Men with diabetes consistently experienced higher cardiovascular mortality than men without diabetes, even when matched for one or more of the major risk factors for CVD, ie, elevated serum cholesterol level, elevated systolic blood pressure level, and cigarette smoking. These data imply that something intrinsic to the diabetic state, perhaps the hyperglycemia itself, confers additional risk of CVD.
|
|

|
|
Figure 1. Age-adjusted cardiovascular disease (CVD) mortality in men with and without diabetes screened for the Multiple Risk Factor Intervention Trial matched on 1 or more of the following risk factors: cholesterol levels, blood pressure levels, and cigarette smoking. From Stamler et al.13 Reprinted with permission of the American Diabetes Association.
|
|
|
In this article, my main purpose is discussion of the evidence that glycemic control in persons with type 2 diabetes will reduce their risk of macrovascular complications, ie, large-vessel atherosclerosis. Two caveats, however, should be noted. First, the importance of treating other cardiovascular risk factors in persons with diabetes should not be minimized. Even if glycemic control contributes to lowering cardiovascular risk, management of dyslipidemia and hypertension and encouragement of smoking cessation remain critically important to further minimize this risk. These topics, however, have been reviewed elsewhere.14-15 Second, the available data on glycemic control and macrovascular complications are inadequate. Although some data are available from clinical trials, a large-scale clinical trial focused specifically on whether "tight" glycemic control lowers macrovascular risk in persons with diabetes has yet to be performed. Until such a trial is performed, an element of uncertainty continues to surround this issue.
GLYCEMIC CONTROL AND MICROVASCULAR COMPLICATIONS
A number of relatively small clinical trials have suggested that intensive insulin regimens leading to tight glycemic control can reduce the risk of microvascular complications.16-18 To confirm these findings, a large and more definitive study, the Diabetes Control and Complications Trial (DCCT), was undertaken.19 A total of 1441 patients with type 1 diabetes were enrolled beginning in 1983; half were randomly allocated to an intensive insulin regimen. The average level of hemoglobin A1c achieved by the intensive therapy group was about 7.0%, compared with 9.0% for the conventional therapy group. The results indicated that, after a mean of 6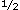 years of follow-up, intensive insulin management was associated with dramatic reductions in the incidence of microvascular complications. The incidence or progression of retinopathy, for example, was reduced by a range of 54% to 76%, and the need for photocoagulation therapy was reduced by 56%. Similarly, the incidence of microalbuminuria was reduced by 39%, the incidence of clinical albuminuria by 54%, and the incidence of clinical neuropathy by 60%. Although these results are impressive, they were not obtained without a cost. In particular, intensive management was associated with a 3-fold increase in hypoglycemic episodes and statistically significant weight gain. years of follow-up, intensive insulin management was associated with dramatic reductions in the incidence of microvascular complications. The incidence or progression of retinopathy, for example, was reduced by a range of 54% to 76%, and the need for photocoagulation therapy was reduced by 56%. Similarly, the incidence of microalbuminuria was reduced by 39%, the incidence of clinical albuminuria by 54%, and the incidence of clinical neuropathy by 60%. Although these results are impressive, they were not obtained without a cost. In particular, intensive management was associated with a 3-fold increase in hypoglycemic episodes and statistically significant weight gain.
The DCCT provides strong—many would say persuasive—evidence that aggressive efforts to control hyperglycemia substantially reduce the risk of microvascular complications. Based on the DCCT results, intensive insulin management has now become the standard of care for type 1 diabetes.20 Moreover, these results likely can be extrapolated to type 2 diabetes, because the pathogenesis of microvascular complications is believed to be essentially the same in both types of diabetes.21 An intensive insulin regimen similar to that used in the DCCT has been tested in a randomized clinical trial of Japanese patients with type 2 diabetes.22 Although the study included only 110 patients, the results were remarkably similar to those of the DCCT. The incidence and progression of retinopathy were reduced in the intensive treatment group vs the conventional insulin therapy group by a range of 56% to 76%. The corresponding reductions for nephropathy were in a range of 64% to 73%. Hypoglycemia and weight gain in this study were minimal.
IS INSULINEMIA OR INSULIN RESISTANCE ATHEROGENIC?
If insulin were atherogenic, then any theoretical benefits of glycemic control on macrovascular complications might be partially or wholly offset by the administration of exogenous insulin or drugs that, like sulfonylureas, stimulate insulin secretion. Presumably, this would not be the case if it were insulin resistance, and not insulinemia, that were atherogenic. Neither would a potential atherogenic effect of insulin be relevant if adequate glycemic control could be achieved with drugs that do not raise endogenous insulin concentrations, such as metformin or troglitazone.
The evidence implicating insulin as a possibly atherogenic substance includes data from animal studies, in vitro studies, and prospective epidemiologic studies. Many of these studies have been summarized by Stout.23 A number of early studies indicated that in cholesterol-fed animals with alloxan-induced diabetes, the expected degree of aortic atherosclerosis that usually follows cholesterol feeding failed to develop unless insulin was simultaneously administered.24-25 In 1 study, injection of insulin directly into 1 femoral artery of a dog with alloxan-induced diabetes resulted in more medial thickening and a higher cholesterol content in the vascular wall than injection of saline into the contralateral femoral artery.26 Insulin also has been shown to cause in vitro proliferation of smooth muscle cells obtained from a number of experimental animals and from humans,23, 27-28 to stimulate sterol synthesis in cultured smooth muscle cells,29 and to stimulate the binding of low-density lipoprotein (LDL) to smooth muscle cells30 and fibroblasts.31 Finally, insulin also stimulates cholesterol synthesis and the binding of LDL to cell membranes in monocytes.32-33 Thus, insulin could theoretically promote atherogenesis by direct action on the arterial wall.
Several prospective epidemiologic studies have tested the hypothesis that the circulating insulin concentration is a cardiovascular risk factor.34-41 None of these studies, however, distinguished between insulin and insulin resistance as the possibly atherogenic factor, because, although circulating insulin concentrations were measured, these concentrations typically reflect underlying insulin resistance in persons without diabetes. Indeed, Reaven,42 who was one of the first to propose that the insulin resistance syndrome was atherogenic, emphasized that the insulin resistance, and not necessarily the circulating insulin concentration, is responsible for the increased cardiovascular risk.43
The early epidemiologic studies34-39 on the role of insulinemia as a possible cardiovascular risk factor gave inconsistent results, and the study designs included a number of defects. For example, most of them did not include measurements of high-density lipoprotein (HDL) cholesterol at baseline, so it was impossible to ascertain whether the presumed insulin effect was independent of the effect of HDL cholesterol, an established cardiovascular risk factor. In addition, many did not adequately distinguish between the effects of insulinemia and those of diabetes, because they failed to adequately exclude persons with diabetes from the analyses. In some studies, diabetes was identified only by self-report, and because glucose tolerance testing was not performed, undiagnosed cases were not identified and excluded. The conventional immunoassays in use when these studies were performed are now known to cross-react with proinsulin and other insulin precursors.44-46 Because it is now known that persons with diabetes have high circulating levels of insulin precursors, and because they also have elevated rates of CVD, a spurious association between "insulinemia" (really proinsulinemia) and CVD could have been produced.
Two prospective epidemiologic studies of insulinemia as a cardiovascular risk factor have dealt with the aforementioned criticisms.40-41 In both studies, HDL cholesterol levels were measured at baseline and adjusted for in the analysis. Also, both studies used specific insulin assays that do not cross-react with proinsulin or other insulin precursors. In the Quebec Cardiovascular Study, 45- to 76-year-old men were followed up prospectively for 5 years.40 Among the men without diabetes, nonfatal myocardial infarction, angina pectoris, or fatal coronary heart disease developed in 91. Specific insulin concentrations at baseline were compared with those in 105 control subjects without diabetes matched for age, body mass index, smoking, and alcohol consumption. An increase of 1.0 SD in the baseline specific insulin concentration was associated with a 70% increased risk of CVD, and this result persisted even after accounting for numerous other risk factors, among them LDL and HDL levels, triglyceride levels, apolipoprotein B levels, body mass index, systolic blood pressure levels, cigarette smoking, alcohol consumption, family history of ischemic heart disease, and the use of 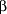 -adrenergic blocking agents or diuretics. Glucose levels, however, were not adjusted for in these analyses. In addition, because hyperinsulinemia usually reflects insulin resistance, the Quebec Cardiovascular Study did not definitively distinguish between insulin resistance and insulin as possible cardiovascular risk factors. -adrenergic blocking agents or diuretics. Glucose levels, however, were not adjusted for in these analyses. In addition, because hyperinsulinemia usually reflects insulin resistance, the Quebec Cardiovascular Study did not definitively distinguish between insulin resistance and insulin as possible cardiovascular risk factors.
In the second study, the British Regional Heart Study,41 5550 men were followed up for 11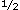 years, and 521 fatal and nonfatal coronary events were observed. Specific insulin concentrations in the top 10% of the distribution at baseline were associated with a 70% increased risk of subsequent CVD, although there was little evidence of a dose-response effect in the lower deciles (Figure 2). The excess risk associated with high specific insulin concentrations remained statistically significant, even after adjustment for a comprehensive panel of conventional cardiovascular risk factors, which included total and HDL cholesterol levels, triglyceride levels, body mass index, systolic blood pressure levels, cigarette smoking, alcohol consumption, physical activity, heart rate, forced expiratory volume in 1 second, and social class. In this study of persons without diabetes, adjustment also was made for the serum glucose level, and the insulin effect was shown to be independent of this potential confounder as well. As in the Quebec Cardiovascular Study, the British Regional Heart Study did not distinguish between insulinemia and insulin resistance as possible cardiovascular risk factors. A further potential weakness of this study was its use of nonfasting specimens for the measurement of glucose and insulin concentrations, but the investigators seem to have handled this issue adequately in their statistical analyses of the data. years, and 521 fatal and nonfatal coronary events were observed. Specific insulin concentrations in the top 10% of the distribution at baseline were associated with a 70% increased risk of subsequent CVD, although there was little evidence of a dose-response effect in the lower deciles (Figure 2). The excess risk associated with high specific insulin concentrations remained statistically significant, even after adjustment for a comprehensive panel of conventional cardiovascular risk factors, which included total and HDL cholesterol levels, triglyceride levels, body mass index, systolic blood pressure levels, cigarette smoking, alcohol consumption, physical activity, heart rate, forced expiratory volume in 1 second, and social class. In this study of persons without diabetes, adjustment also was made for the serum glucose level, and the insulin effect was shown to be independent of this potential confounder as well. As in the Quebec Cardiovascular Study, the British Regional Heart Study did not distinguish between insulinemia and insulin resistance as possible cardiovascular risk factors. A further potential weakness of this study was its use of nonfasting specimens for the measurement of glucose and insulin concentrations, but the investigators seem to have handled this issue adequately in their statistical analyses of the data.
|
|

|
Figure 2. British Regional Heart Study. Coronary heart disease event rates after 11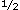 years of follow-up according to the quintile of serum specific insulin concentration at baseline. The numbers in parentheses are the numbers of men in each quintile and the numbers above the bars are the number of men experiencing an event. Reprinted with permission of the American Journal of Epidemiology, Johns Hopkins University School of Hygiene and Public Health (Perry et al41). years of follow-up according to the quintile of serum specific insulin concentration at baseline. The numbers in parentheses are the numbers of men in each quintile and the numbers above the bars are the number of men experiencing an event. Reprinted with permission of the American Journal of Epidemiology, Johns Hopkins University School of Hygiene and Public Health (Perry et al41).
|
|
|
In summary, the animal and in vitro data suggesting that insulin is atherogenic are weak. The epidemiologic data do not distinguish between insulin and insulin resistance as cardiovascular risk factors, because most persons with hyperinsulinemia also have insulin resistance.
HYPERGLYCEMIA AS A RISK FACTOR FOR ATHEROSCLEROSIS
The aforementioned epidemiologic studies relate primarily to the effects of endogenous insulin in euglycemic persons, ie, persons without diabetes. If it could be shown that hyperglycemia increases cardiovascular risk, one might then argue that glycemic control with exogenous insulin in persons with diabetes could reduce cardiovascular risk even if endogenous insulin were atherogenic in persons without diabetes. In other words, the benefits of glycemic control might outweigh any theoretical atherogenic effects of insulin.
Hyperglycemia is associated with glycosylation of virtually every protein in the body. Many of these biochemical changes could contribute to atherogenesis. For example, advanced glycation end products induce excessive cross-linking of collagen and other extracellular matrix proteins in many tissues, including the vascular wall.47-48 This excessive cross-linking causes covalent trapping of LDLs, which may then accumulate in vascular walls.48 In addition, apolipoprotein B, a component of LDL, can be modified by advanced glycation end products.49 Such advanced glycation end product–modified LDL has been shown to have a prolonged half-life,50 which, combined with trapping of the lipoprotein in vascular walls, could lead to increased oxidation of LDL. Oxidized LDL is believed to be more atherogenic than native LDL.51
Empirically, early studies among persons without diabetes suggested that glycemia was a weak and inconsistent risk factor for CVD.52 Park et al53 reported, however, that glycohemoglobin predicted mortality due to CVD and ischemic heart disease in women without diabetes, although not in men without diabetes. Glycohemoglobin values in the fifth quintile at baseline were associated with increased risk, but there was no graded response in the lower 4 quintiles. Thus, a threshold effect was suggested. This effect was independent of body mass index, systolic blood pressure levels, LDL and HDL cholesterol levels, triglyceride levels, cigarette smoking, the use of antihypertensives, and, in women, of the use of estrogens. Unfortunately, the simultaneous effect of insulinemia in this cohort was not reported. In contrast to glycohemoglobin, neither fasting nor post–oral-load glucose values predicted mortality due to CVD or ischemic heart disease in either sex. The authors argued that the reason glycohemoglobin seemed to be a risk factor (at least in women) was that it represented the integrated level of glycemia over many weeks, rather than reflecting a single point in time, as is the case with plasma glucose measurements. Even so, the effect was seen only in women and in the fifth quintile of the glycohemoglobin distribution with no dose effect in the 4 lower quintiles.
Impaired glucose tolerance is generally regarded as a cardiovascular risk factor, but that is probably because it forms part of the cluster of metabolic and physiologic factors that constitute the insulin resistance syndrome (syndrome X), consisting of obesity, dyslipidemia, and hypertension, each of which is a cardiovascular risk factor.42 It is doubtful that impaired glucose tolerance is associated with increased cardiovascular risk once these other risk factors are considered.
Among persons with diabetes, early, mainly cross-sectional, studies did not support the concept that the degree of hyperglycemia influenced the risk of macrovascular complications. For example, in the WHO (World Health Organization) Multinational Study of diabetic complications, no relationship was found between plasma glucose levels and Q waves on electrocardiograms indicating myocardial infarction.54 More recent studies, however, have suggested an opposite conclusion. Kuusisto et al55 reported a 3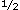 -year follow-up of elderly Finnish persons with diabetes (65-74 years old). In that study, glycohemoglobin and the duration of diabetes were associated in a graded fashion with fatal coronary heart disease (CHD) and fatal and nonfatal CHD combined (Figure 3) even after adjusting for total and HDL cholesterol levels, triglyceride levels, systolic blood pressure levels, cigarette smoking, and previous myocardial infarction. These classic cardiovascular risk factors did not independently predict CHD once the effect of glycohemoglobin was considered. In addition, fasting and post–oral-load plasma glucose levels failed to predict CHD in this study, which may reflect the instantaneous nature of plasma glucose measurements compared with glycohemoglobin. Unfortunately, the effect of insulin on CHD was not considered. The Framingham Study also reported that glycohemoglobin, but not plasma glucose, was cross-sectionally associated with CVD in women, although not in men.56 This effect was independent of the total-to-HDL cholesterol ratio, hypertension, cigarette smoking, body mass index, diabetes, and treatment with -year follow-up of elderly Finnish persons with diabetes (65-74 years old). In that study, glycohemoglobin and the duration of diabetes were associated in a graded fashion with fatal coronary heart disease (CHD) and fatal and nonfatal CHD combined (Figure 3) even after adjusting for total and HDL cholesterol levels, triglyceride levels, systolic blood pressure levels, cigarette smoking, and previous myocardial infarction. These classic cardiovascular risk factors did not independently predict CHD once the effect of glycohemoglobin was considered. In addition, fasting and post–oral-load plasma glucose levels failed to predict CHD in this study, which may reflect the instantaneous nature of plasma glucose measurements compared with glycohemoglobin. Unfortunately, the effect of insulin on CHD was not considered. The Framingham Study also reported that glycohemoglobin, but not plasma glucose, was cross-sectionally associated with CVD in women, although not in men.56 This effect was independent of the total-to-HDL cholesterol ratio, hypertension, cigarette smoking, body mass index, diabetes, and treatment with 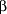 -adrenergic blocking agents or thiazides. The effect of insulin, however, was not assessed. In a study of newly diagnosed nonelderly Finnish persons with diabetes, plasma glucose and glycohemoglobin were predictive of CVD independently of other cardiovascular risk factors, including LDL and HDL cholesterol levels, systolic blood pressure levels, cigarette smoking, body mass index, and fasting insulin concentrations.57 In the Wisconsin Epidemiologic Study of Diabetic Retinopathy, 2990 persons with diabetes, representing a probability sample of all persons with diabetes in an 11-county area in southern Wisconsin, were followed up for 10 years.58 A 1% increase in glycohemoglobin at baseline was associated with an 18% increase in mortality due to ischemic heart disease in persons with early-onset diabetes (probably type 1 diabetes) and a 10% increase in mortality due to ischemic heart disease in persons with late-onset diabetes (probably type 2 diabetes). Although less than the relative risks associated with microvascular complications, these relative risks for macrovascular complications were, nevertheless, statistically significant. -adrenergic blocking agents or thiazides. The effect of insulin, however, was not assessed. In a study of newly diagnosed nonelderly Finnish persons with diabetes, plasma glucose and glycohemoglobin were predictive of CVD independently of other cardiovascular risk factors, including LDL and HDL cholesterol levels, systolic blood pressure levels, cigarette smoking, body mass index, and fasting insulin concentrations.57 In the Wisconsin Epidemiologic Study of Diabetic Retinopathy, 2990 persons with diabetes, representing a probability sample of all persons with diabetes in an 11-county area in southern Wisconsin, were followed up for 10 years.58 A 1% increase in glycohemoglobin at baseline was associated with an 18% increase in mortality due to ischemic heart disease in persons with early-onset diabetes (probably type 1 diabetes) and a 10% increase in mortality due to ischemic heart disease in persons with late-onset diabetes (probably type 2 diabetes). Although less than the relative risks associated with microvascular complications, these relative risks for macrovascular complications were, nevertheless, statistically significant.
|
|

|
Figure 3. The 3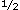 -year incidence of mortality due to coronary heart disease (CHD) (top) and CHD events (bottom) in persons with type 2 diabetes according to tertile of glycohemoglobin level. Reprinted with permission from the American Diabetes Association (Kuusisto et al55). -year incidence of mortality due to coronary heart disease (CHD) (top) and CHD events (bottom) in persons with type 2 diabetes according to tertile of glycohemoglobin level. Reprinted with permission from the American Diabetes Association (Kuusisto et al55).
|
|
|
These studies indicate that the level of hyperglycemia in persons with diabetes influences the risk of macrovascular and microvascular complications, perhaps through some of the aforementioned mechanisms. Thus, tight control of diabetes with insulin has the potential of lowering cardiovascular risk, even if endogenous insulin is atherogenic in persons without diabetes.
EFFECT OF EXOGENOUS INSULIN ON CARDIOVASCULAR RISK FACTORS AND CVD END POINTS
For lipids and lipoproteins, substantial literature suggests that exogenous insulin lowers very low density lipoprotein cholesterol and triglyceride levels.59-60 In addition, some studies show a shift toward higher levels of HDL2 and lower levels of HDL3 with little or no effect on total HDL cholesterol levels.60 This shift may be beneficial, because HDL2 is believed to be more antiatherogenic than HDL3.61 Almost all studies on the effects of exogenous insulin on lipids and lipoproteins are relatively small (<100 patients), of short duration (weeks to months), and without randomized controls. An important exception is the DCCT. This study included randomized controls, and lipid responses were assessed for up to 9 years. The results, shown in Figure 4, indicate that intensive insulin management is associated with modest reductions in total and LDL cholesterol and triglyceride levels, but no change in HDL cholesterol levels.62 Blood pressure levels were also similar in the 2 treatment arms.
|
|

|
|
Figure 4. Mean total, low-density lipoprotein (LDL), and high-density lipoprotein (HDL) cholesterol and triglyceride levels in intensively treated (solid line) and conventionally treated (broken line) patients in the Diabetes Control and Complications Trial in the primary prevention cohort (left) and the secondary prevention cohort (right). Reprinted with permission of the publisher from reference 62, copyright 1995, by Excerpta Medica Inc.62
|
|
|
The feasibility trial of the Veterans Affairs Cooperative Study on Glycemic Control and Complications in Type II Diabetes (VA CSDM) also studied lipid and lipoprotein responses during a 27-month period in patients randomized to intensive vs standard therapy with exogenous insulin.63 In this study involving 153 patients, no differences in lipid or lipoprotein levels between the 2 treatment groups emerged during the trial, despite improved glycemic control in the intensively managed group (glycohemoglobins about 2% lower than in the standard therapy group).
For persons whose diabetes can be controlled with oral antidiabetic agents, the possibility exists of using agents that do not raise insulin concentrations. For example, metformin controls hyperglycemia with minimal or no changes in circulating insulin concentrations.64 It also lowers total and LDL cholesterol and triglyceride levels.64 The insulin-sensitizing agent, troglitazone, is a member of a new class of agents known as thiazolidinediones.65 This agent has been reported to reduce insulin concentrations and insulin resistance in persons with66 and without diabetes.65 Furthermore, it has been reported to lower blood pressure65 and triglyceride levels.67 In a randomized comparison of glyburide and troglitazone involving 154 patients followed up for 48 weeks, troglitazone significantly lowered insulin and triglyceride levels and raised HDL levels. Glyburide, by contrast, was associated with a nonsignificant rise in insulin and HDL levels and a nonsignificant decline in triglyceride levels.68 The favorable lipid changes associated with troglitazone were achieved when this drug was used alone, not in combination with insulin as is currently recommended. Moreover, troglitazone was also associated with a modest rise in total and LDL cholesterol levels, whereas glyburide was not.68
Several randomized clinical trials have compared cardiovascular end points in patients receiving different antidiabetic regimens. Unfortunately, none of these studies can be regarded as definitive, generally because of an inadequate sample or few events. The earliest of these trials is the University Group Diabetes Program.69 In this study, approximately 200 patients with type 2 diabetes were randomized to the following treatment regimens: placebo, tolbutamide, a fixed insulin dose, and a variable insulin dose titrated to maintain glycemic control. One result of this study was that cardiovascular mortality was significantly higher in the group randomized to tolbutamide compared with the other 3 groups. This finding, not surprisingly, stirred considerable controversy—which, unfortunately, diverted attention from other important study results. Cardiovascular mortality, for example, was similar in the other 3 groups; neither of the insulin regimens increased or decreased cardiovascular mortality relative to placebo. Perhaps the atherogenic effects of insulin were precisely balanced by its beneficial effects mediated through glycemic control, resulting in less atherogenesis-provoking glycosylation of proteins. If this interpretation is correct, and if insulin therapy has no beneficial or adverse effect on macrovascular complications, then it would still be appropriate to conclude that tight glycemic control is indicated because of its proven ability to reduce microvascular complications as demonstrated by the DCCT.19
The DCCT also reported results for macrovascular complications.62 Because the patients in the DCCT were young (13-39 years old at entry), relatively few macrovascular events were recorded. There were only 3 cardiovascular deaths, 2 in the intensively treated group and 1 in the conventionally treated group. In addition, 13 patients in the conventional group, but only 1 in the intensively treated group, experienced nonfatal cardiovascular events, which included myocardial infarction, angina pectoris, and coronary artery bypass grafting. Although not statistically significant, these results are encouraging.
Unfortunately, the results of the VA CSDM feasibility study involving 153 subjects are less encouraging. In this study, 78 patients were randomized to standard care and 75 to intensive glycemic management. Thirty-five major cardiovascular events (ie, myocardial infarction, congestive heart failure, stroke, amputation, angina, angioplasty, coronary bypass surgery, or cardiovascular death) were recorded during a 27-month period in 24 patients (32%) receiving intensive insulin therapy compared with only 26 events in 16 patients (21%) receiving standard therapy.70 The incidence of major cardiovascular events was thus 52% higher in the intensive therapy group than in the standard therapy group, although this difference was only of borderline significance (P=.10).
Finally, the Diabetes Mellitus Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) Study, a randomized controlled trial, reported that insulin-glucose infusions administered to persons with diabetes during the immediate postmyocardial infarction period, followed by multiple subcutaneous insulin injections for 3 months or more, improved the prognosis during the ensuing 12 months.71 Whether this benefit resulted from the immediate insulin-glucose infusion or from improved long-term control is unclear because the study design did not distinguish between these possibilities. Of note is the fact that at 3 months of follow-up, the glucose and glycohemoglobin levels were only slightly lower in the group that had received the infusion, and there was no difference in glycohemoglobin at 1 year of follow-up.
In addition, in the UK (United Kingdom) Prospective Study,72 patients with type 2 diabetes have been randomized to several different antidiabetic regimens, including metformin. This study is still in progress and results have not yet been reported.
The results from randomized clinical trials on the effects of glycemic control on cardiovascular end points are inconsistent, perhaps because they are based on relatively few cases. A large-scale definitive trial to resolve this question is urgently needed.
CONCLUSIONS
There is now overwhelming evidence that tight control of hyperglycemia in persons with type 1 diabetes will dramatically reduce the incidence of microvascular complications. There is also highly suggestive evidence that this conclusion can be extended to persons with type 2 diabetes. One concern that has been raised about the implementation of tight control with intensive insulin regimens is the possibility that insulin is atherogenic and might paradoxically increase the risk of macrovascular complications. Many prospective epidemiologic studies have suggested that the endogenous insulin concentration is a risk factor for CVD in persons without diabetes, although these studies do not distinguish between insulin and insulin resistance as the factor associated with increased cardiovascular risk. The level of glycemia does not seem to be a risk factor for CVD in persons without diabetes, whereas it probably is a cardiovascular risk factor in persons with diabetes. The mechanism whereby hyperglycemia enhances cardiovascular risk in persons with diabetes may involve glycosylation of various proteins, such as apolipoprotein B and collagen in vascular walls. Thus, glycemic control in persons with diabetes, whether by intensive insulin management or other modalities, has the potential of lowering cardiovascular risk—a benefit not available to euglycemic persons. Even if endogenous insulin were atherogenic, a proposition that is by no means proved, controlling hyperglycemia with exogenous insulin could theoretically offset these adverse effects by reducing atherogenesis-provoking glycosylation. Of course, this concept presupposes that the hyperglycemia is, in fact, controlled—which often is not the case. Otherwise, the presumed atherogenic effects of insulin would act unopposed. In addition, insulin-sensitizing agents, such as metformin and troglitazone, could, if they adequately controlled the glycemia, offer a further advantage because they do not change or reduce endogenous insulin concentrations. Clinical trials on the effects of glycemic control on the macrovascular complications of diabetes are limited and do not provide definitive answers. There is a great need for a large-scale definitive trial to resolve this highly relevant clinical question. Finally, in addition to adequate glycemic control in persons with diabetes, aggressive management of other cardiovascular risk factors (eg, dyslipidemia, hypertension, and cigarette smoking) is also indicated.
AUTHOR INFORMATION
Accepted for publication June 17, 1997.
Reprints: Michael P. Stern, MD, Division of Clinical Epidemiology, Department of Medicine, University of Texas Health Science Center at San Antonio, 7703 Floyd Curl Dr, San Antonio, TX 78284-7873 (e-mail: stern{at}uthscsa.edu).
From the Division of Clinical Epidemiology, Department of Medicine, University of Texas Health Science Center at San Antonio.
REFERENCES
1. Geiss LS, Herman WH, Smith PJ. Mortality in non–insulin-dependent diabetes. In: Harris MI, ed. Diabetes in America. 2nd ed. Bethesda, Md: National Institute of Diabetes and Digestive and Kidney Diseases; 1995:233-257. National Institutes of Health publication 95-1468.
2. American Diabetes Association. Direct and Indirect Costs of Diabetes in the United States in 1992. Alexandria, Va: American Diabetes Association; 1993.
3. Kannel WB, McGee DL. Diabetes and glucose tolerance as risk factors for cardiovascular disease: the Framingham Study. Diabetes Care. 1979;2:120-126.
FREE FULL TEXT
4. Barrett-Connor EL, Cohn BA, Wingard DL, Edelstein SL. Why is diabetes mellitus a stronger risk factor for fatal ischemic heart disease in women than in men? the Rancho Bernardo Study. JAMA. 1991;265:627-631.
FREE FULL TEXT
5. Tull ES, Roseman JM. Diabetes in African Americans. In: Harris MI, ed. Diabetes in America. 2nd ed. Bethesda, Md: National Institute of Diabetes and Digestive and Kidney Diseases; 1995:613-630. National Institutes of Health publication 95-1468.
6. Stern MP, Mitchell BD. Diabetes in Hispanic Americans. In: Harris MI, ed. Diabetes in America. 2nd ed. Bethesda, Md: National Institute of Diabetes and Digestive and Kidney Diseases; 1995:631-659. National Institutes of Health publication 95-1468.
7. Gohdes D. Diabetes in North American Indians and Alaska Natives. In: Harris MI, ed. Diabetes in America. 2nd ed. Bethesda, Md: National Institute of Diabetes and Digestive and Kidney Diseases; 1995:683-701. National Institutes of Health publication 95-1468.
8. Nelson RG, Sievers ML, Knowler WC, et al. Low incidence of fatal coronary heart disease in Pima Indians despite high prevalence of non–insulin-dependent diabetes. Circulation. 1990;81:987-995.
FREE FULL TEXT
9. Howard BV, Lee ET, Cowan LD, et al. Coronary heart disease prevalence and its relation to risk factors in American Indians: the Strong Heart Study. Am J Epidemiol. 1995;142:254-268.
FREE FULL TEXT
10. Sorlie PD, Backlund E, Johnson NJ, Rogot E. Mortality by Hispanic status in the United States. JAMA. 1993;270:2464-2468.
FREE FULL TEXT
11. Wei M, Mitchell BD, Haffner SM, Hazuda HP, Stern MP. The effects of cigarette smoking, diabetes, high cholesterol, and hypertension on all-cause mortality and cardiovascular disease mortality in Mexican Americans: the San Antonio Heart Study. Am J Epidemiol. 1996;144:1058-1065.
FREE FULL TEXT
12. Nichaman MZ, Wear ML, Goff DC, Labarthe DR. Hospitalization rates for myocardial infarction among Mexican Americans and non-Hispanic whites: the Corpus Christi Heart Project. Ann Epidemiol. 1993;3:42-48.
PUBMED
13. Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-year cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care. 1993;16:434-444.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
14. Haffner SM. Management of dyslipidemia in adults with diabetes: ADA technical review. Diabetes Care. 1998;21:160-178.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
15. American Diabetes Association. Consensus statement: treatment of hypertension in diabetes. Diabetes Care. 1993;16:1394-1401.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
16. Kroc Collaborative Study Group. Blood glucose control and the evolution of diabetic retinopathy and albuminuria: a preliminary multicenter trial. N Engl J Med. 1984;311:365-372.
WEB OF SCIENCE
| PUBMED
17. Brinchmann-Hansen O, Dahl-Jorgensen K, Hanssen KF, Sanvik L. The response of diabetic retinopathy to 41 months of multiple insulin injections, insulin pumps, and conventional insulin therapy. Arch Ophthalmol. 1988;106:1242-1246.
FREE FULL TEXT
18. Reichard P, Nilsson B-Y, Rosenqvist U. The effect of long-term intensified insulin treatment on the development of microvascular complications of diabetes mellitus. N Engl J Med. 1993;329:304-309.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
19. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977-986.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
20. American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care. 1997;20(suppl 1):S5-S13.
21. American Diabetes Association. Implications of the Diabetes Control and Complications Trial. Diabetes Care. 1993;16:1517-1520.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
22. Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non–insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract. 1995;28:103-117.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
23. Stout RW. Insulin and atheroma, 20-year perspective. Diabetes Care. 1990;13:631-654.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
24. McGill HC Jr, Holman RL. The influence of alloxan diabetes on cholesterol atheromatosis in the rabbit. Proc Soc Exp Biol Med. 1949;72:72-73.
25. Lehner NDM, Clarkson TB, Lofland HB. The effect of insulin deficiency, hypothyroidism, and hypertension on atherosclerosis in the squirrel monkey. Exp Mol Pathol. 1971;15:230-244.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
26. Cruz AB Jr, Amatuzio DS, Grande F, Hay LJ. Effect of intra-arterial insulin on tissue cholesterol and fatty acids in alloxan-diabetic dogs. Circ Res. 1961;9:39-43.
FREE FULL TEXT
27. Stout RW, Bierman EL, Ross R. Effect of insulin on the proliferation of cultured primate arterial smooth muscle cells. Circ Res. 1975;36:319-327.
FREE FULL TEXT
28. Pfeifle B, Ditschuneit H. Effect of insulin on growth of cultured human arterial smooth muscle cells. Diabetologia. 1977;20:155-158.
29. Stout RW. The effect of insulin and glucose on sterol synthesis in cultured rat arterial smooth muscle cells. Atherosclerosis. 1977;27:271-278.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
30. Young IR, Stout RW. Effects of insulin and glucose on the cells of the arterial wall: interaction of insulin with dibutyryl cyclic AMP and low density lipoprotein in arterial cells. Diabetes Metab. 1987;13:301-306.
WEB OF SCIENCE
31. Oppenheimer MJ, Sundquist K, Bierman EL. Down-regulation of high-density lipoprotein receptor in human fibroblasts by insulin and IGF-I. Diabetes. 1989;38:117-122.
ABSTRACT
32. Krone W, Greten H. Evidence for post-transcriptional regulation by insulin of 3-hydroxy-3-methylglutaryl coenzyme A reductase and sterol synthesis in human mononuclear leukocytes. Diabetologia. 1984;26:366-369.
WEB OF SCIENCE
| PUBMED
33. Krone W, Nagele H, Behnke B, Greten H. Opposite effects of insulin and catecholamines on LDL-receptor activity in human mononuclear leukocytes. Diabetes. 1988;37:1386-1391.
ABSTRACT
34. Pyörälä K. Relationship of glucose tolerance and plasma insulin to the incidence of coronary heart disease: results from two population studies in Finland. Diabetes Care. 1979;2:131-141.
FREE FULL TEXT
35. Ducimetiere P, Eschwege E, Papoz L, Richard JL, Claude JR, Rosselin G. Relationship of plasma insulin levels to the incidence of myocardial infarction and coronary heart disease mortality in a middle-aged population. Diabetologia. 1980;19:205-210.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
36. Welborn TA, Wearne K. Coronary heart disease incidence and cardiovascular mortality in Busselton with reference to glucose and insulin concentrations. Diabetes Care. 1979;2:154-160.
FREE FULL TEXT
37. Welin L, Eriksson H, Larsson B, Ohlson LO, Svärdsudd K, Tibblin G. Hyperinsulinemia is not a major coronary risk factor in elderly men: the study of men born in 1913. Diabetologia. 1992;35:766-770.
WEB OF SCIENCE
| PUBMED
38. Orchard TJ, Eichner J, Kuller LH, Becker DJ, McCallum LM, Grandits GA. Insulin as a predictor of coronary heart disease: interaction with apolipoprotein E phenotype: a report from the Multiple Risk Factor Intervention Trial. Ann Epidemiol. 1994;4:40-45.
PUBMED
39. Ferrara A, Barrett-Connor EL, Edelstein SL. Hyperinsulinemia does not increase the risk of fatal cardiovascular disease in elderly men or women without diabetes: the Rancho Bernardo Study, 1984-1991. Am J Epidemiol. 1994;140:857-869.
FREE FULL TEXT
40. Deprés J-P, Lamarche B, Mauriege P, et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med. 1996;334:952-957.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
41. Perry IJ, Wannamethee SG, Whincup PH, Shaper AG, Walker MK, Alberti KGMM. Serum insulin and incident coronary heart disease in middle-aged British men. Am J Epidemiol. 1996;144:224-234.
FREE FULL TEXT
42. Reaven GM. Banting Lecture 1988: role of insulin resistance in human disease. Diabetes. 1988;37:1595-1607.
ABSTRACT
43. Reaven GM, Laws A. Insulin resistance, compensatory hyperinsulinemia, and coronary heart disease. Diabetologia. 1994;37:948-952.
WEB OF SCIENCE
| PUBMED
44. Temple RC, Clark PM, Nagi DK, Schneider AE, Yudkin JS, Hales CN. Radioimmunoassay may overestimate insulin in non–insulin-dependent diabetics. Clin Endocrinol (Oxf). 1990;32:689-693.
PUBMED
45. Saad MF, Kahn SE, Nelson RG, et al. Disproportionately elevated proinsulin in Pima Indians with non–insulin-dependent diabetes mellitus. J Clin Endocrinol Metab. 1990;70:1247-1253.
FREE FULL TEXT
46. Haffner SM, Bowsher RR, Mykkänen L, et al. Proinsulin and specific insulin concentration in high- and low-risk populations for NIDDM. Diabetes. 1994;43:1490-1493.
ABSTRACT
47. Brownlee M. Glycation products and the pathogenesis of diabetic complications. Diabetes Care. 1992;15:1835-1843.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
48. Vlassara H, Bucala R. Recent progress in advanced glycation and diabetic vascular disease: role of advanced glycation end product receptors. Diabetes. 1996;45(suppl 3):S65-S66.
49. Bucala R, Makita Z, Koschinsky T, Cerami A. Lipid advanced glycation: pathway for lipid oxidation in vivo. Proc Natl Acad Sci U S A. 1993;90:6434-6438.
FREE FULL TEXT
50. Bucala R, Makita Z, Vega G, Grundy S, Koschinsky T, Cerami A, Vlassara H. Modification of low-density lipoprotein by advanced glycosylation end products contributes to the dyslipidemia of diabetes and renal insufficiency. Proc Natl Acad Sci U S A. 1994;91:9441-9445.
FREE FULL TEXT
51. Steinberg D, Parthasarathy S, Carew TE, Khou JC, Witztum JL. Beyond cholesterol: modifications of low density lipoprotein that increase its atherogenicity. N Engl J Med. 1989;320:913-924.
52. Stamler R, ed, Stamler J, ed. Asymptomatic hyperglycaemia and coronary heart disease: a series of papers by the International Collaborative Group, based on studies in fifteen populations. J Chronic Dis. 1979;32:683-691.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
53. Park S, Barrett-Connor E, Wingard DL, Shan J, Edelstein S. GHb is a better predictor of cardiovascular disease than fasting or postchallenge plasma glucose in women without diabetes: the Rancho Bernardo Study. Diabetes Care. 1996;19:450-456.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
54. West KM, Ahuja MMS, Bennett PH, et al. The role of circulating glucose and triglyceride concentrations and their interactions with other "risk factors" as determinants of arterial disease in nine diabetic population samples from the WHO Multinational Study. Diabetes Care. 1983;6:361-369.
FREE FULL TEXT
55. Kuusisto J, Mykkänen L, Pyörälä K, Laakso M. NIDDM and its metabolic control predict coronary heart disease in elderly subjects. Diabetes. 1994;43:960-967.
ABSTRACT
56. Singer DE, Nathan DM, Anderson ICM, Wilson PWF, Evans JC. Association of GHbA1c with prevalent cardiovascular disease in the original cohort of the Framingham Heart Study. Diabetes. 1992;41:202-208.
ABSTRACT
57. Uusitupa MI, Niskanen LK, Siitonen O, Voutilainen E, Pyörälä K. Ten-year cardiovascular mortality in relation to risk factors and abnormalities in lipoprotein composition in type 2 (non–insulin-dependent) diabetic and non-diabetic subjects. Diabetologia. 1993;36:1175-1184.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
58. Klein R. Hyperglycemia and microvascular and macrovascular disease in diabetes. Diabetes Care. 1995;18:258-268.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
59. Henry RR, Gumbiner B, Ditzler T, Wallace P, Lyon R, Glauber HS. Intensive conventional insulin therapy for type II diabetes: metabolic effects during a 6-month outpatient trial. Diabetes Care. 1993;16:21-31.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
60. Taskinen MR, Kuusi T, Helve E, Nikkila EA, Yki-Jarvinen H. Insulin therapy induces antiatherogenic changes of serum lipoproteins in noninsulin-dependent diabetes. Arteriosclerosis. 1988;8:168-177.
FREE FULL TEXT
61. Barter PJ, Rye K-A. High density lipoproteins and coronary heart disease. Atherosclerosis. 1996;121:1-12.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
62. Diabetes Control and Complications Trial Research Group. Effect of intensive diabetes management on macrovascular events and risk factors in the Diabetes Control and Complications Trial. Am J Cardiol. 1995;75:894-903.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
63. Abraira C, Colwell JA, Nuttall FQ, et al. Veterans Affairs Cooperative Study on Glycemic Control and Complications in Type II Diabetes (VA CSDM): results of the feasibility trial. Diabetes Care. 1995;18:1113-1123.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
64. DeFronzo RA, Goodman AM The Multicenter Metformin Study Group. Efficacy of metformin in patients with non–insulin-dependent diabetes mellitus. N Engl J Med. 1995;333:541-549.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
65. Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone. N Engl J Med. 1994;331:1188-1193.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
66. Suter SL, Nolan JJ, Wallace P, Gumbiner B, Olefsky JM. Metabolic effects of new oral hypoglycemic agent CS-045 in NIDDM subjects. Diabetes Care. 1992;15:193-203.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
67. Iwamoto Y, Kosaka K, Kuzuya T, Akanuma V, Shigeta Y, Kaneko T. Effects of troglitazone: a new hypoglycemic agent in patients with NIDDM poorly controlled by diet therapy. Diabetes Care. 1996;19:151-156.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
68. Ghazzi MN, Perez JE, Antonucci TK, et al. Cardiac and glycemic benefits of troglitazone treatment in NIDDM. Diabetes. 1997;46:433-439.
ABSTRACT
69. University Group Diabetes Program. A study of the effects of hypoglycemic agents on vascular complications in patients with adult-onset diabetes, II: mortality results. Diabetes. 1970;19(suppl 2):785-830.
70. Abraira C, Colwell J, Nuttall F, et al. Cardiovascular events and correlates in the Veterans Affairs Diabetes Feasibility Trial. Arch Intern Med. 1997;157:181-188.
FREE FULL TEXT
71. Malmberg K, Rydén L, Efendic S, et al. Randomized trial of insulin-glucose infusion followed by subcutaneous insulin treatment in diabetic patients with acute myocardial infarction (DIGAMI Study): effects on mortality at 1 year. J Am Coll Cardiol. 1995;26:57-65.
ABSTRACT
72. UK Prospective Study of Therapies of Maturity-Onset Diabetes, I: effect of diet, sulfonylurea, insulin or biguanide therapy on fasting plasma glucose and body weight over one year. Diabetologia. 1983;24:404-421.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
Divergent effects of low-O2 tension and iloprost on ATP release from erythrocytes of humans with type 2 diabetes: implications for O2 supply to skeletal muscle
Sprague et al.
Am. J. Physiol. Heart Circ. Physiol. 2010;299:H566-H573.
ABSTRACT
| FULL TEXT
Association between Glycosylated Hemoglobin Level and Cardiovascular and All-Cause Mortality in Type 1 Diabetes
Shankar et al.
Am J Epidemiol 2007;166:393-402.
ABSTRACT
| FULL TEXT
|